Close Vs. Seal? Maude Database Analysis Of The Angio-sealÖ And Perclose Proglide« System
Joo Pok, BS, Kedar S. Lavingia, MD, Michael F. Amendola, MD, MEHP.
Virginia Commonwealth University, RIchmond, VA, USA.
OBJECTIVES: The Manufacturer and User Facility Device Experience (MAUDE) Database was established by the FDA to allow voluntary reporting of adverse outcomes with medical devices. We set out to compare rates of adverse vascular events between two commonly used femoral arterial closure systems: Angio-SealÖ (AS) and Perclose ProGlide« (PP).
METHODS: The MAUDE database was accessed to compile a database of reports related to AS and PP from July 2018 to October 2019. Three categories of ōHematomaö, ōcut-downö, and ōmanual compressionö were key words highlighted in the event description of the database entries to search for the adverse events. Case report numbers and event descriptions were reviewed to remove duplicate reports and those with negative findings. Average number of device issues was also included based on the total number of unique device issues reported per entry. Criteria for determining unique device issued included any reported device or device component malfunction. Analysis was undertaken with *FisherÆs Exact and åtwo-tail t-test.
RESULTS: A total of 2,930 total reports were examined (1465 reports for each closure device), of which 568 were AG and 431 PP for adverse events. Comparing the three categories of complications in TABLE 1. 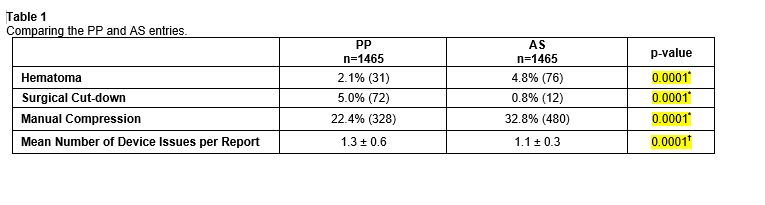 69.7% (n=396) PP and 98.8% (n=426) AS entries stated the access French size.
69.7% (n=396) PP and 98.8% (n=426) AS entries stated the access French size. 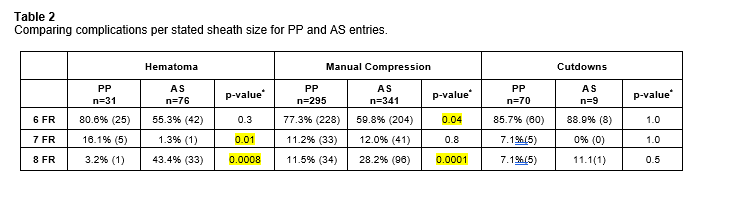
CONCLUSIONS: Overall, there were significantly more hematomas and manual compressions needed to achieve hemostasis following AS usage compared to PP, whereas PP required more surgical cut-downs. PP, however had more device issues per report compared to AS. We excluded patients for comparison in the PP group with larger than 8fr to compare to AS, as many physicians will use multiple in a preclose fashion. AS had a higher rate of hematomas and manual compression at the 8Fr arteriotomy. Interestingly, at a 6fr sheath size, there were more required manual compressions with the PP device, suggesting that PP in smaller more calcified vessels are not suitable candidates for this device whilst with larger arteriotomies, the PP provides for more reliable hemostasis.
Back to 2021 ePosters
