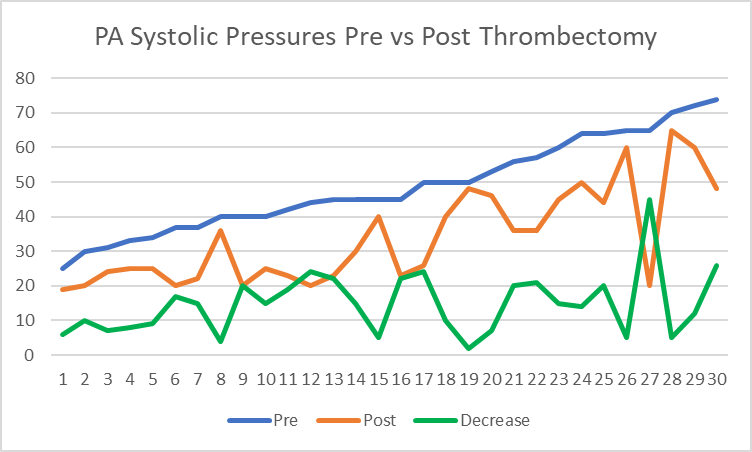
Background: Few single center studies have been published showing clinical benefits of endovascular intervention using mechanical suction thrombectomy for P.E. without systemic/catheter lytic therapy. This study will analyze our initial experience and clinical outcomes in the treatment of P.E. using Inari FlowTriever device. Patient Population/Methods: A single center retrospective study of 50 consecutive patients with acute P.E. were treated with the INARI FlowTriever between January 2019 to June 2023. All patients were submassive P.E. with right ventricular strain (RV/LV ratio >0.9) and requiring oxygen therapy or having increase in oxygen therapy from baseline. Procedural and clinical success were evaluated along with procedural related complications at a mean follow up of 8 months. Results: All patients were stratified as intermediate risk PE with a mean age of 68 years. Right ventricular dilation was present in 100% of patients with a mean RV/LV ratio of 1.55. Mean pre-op Troponin was 434, and mean BNP was 352. Procedural success as defined by SIR guidelines was achieved in 100% of patients. All cause in hospital mortality was 2% without device related mortality, or associated morbidity. Significant improvement in FiO2 was noted from the mean pre-procedural 40.6% to 28.3% at 24 hours post-op (p <0.0001), and proportion of patients on room air increased from 0% pre procedure to 94% at 48 hours. The mean pulmonary artery pressure improved from 47 to 34mmHg (p<0.0001), with 97% of patients had decrease in their pre-op pulmonary pressure. (Figure 1) (Table I) In late follow-up, 36/38 (94.4%) of patients were at or improved from baseline for oxygenation, 34/38 (89%) for physical activity and 37/38 (97%) for breathing status. Using the modified Medical Research Council Dyspnea Score, 93% of patients reported a pre-op score of 4 (highest) while 70% reported scores of 0 (normal) and or 1 post intervention with 100% showing improvement. Conclusion: Our study demonstrates both safety and effectiveness in using the INARI FlowTriever for patients with submassive acute pulmonary embolism.
| Variable | Mean | Median | Std Dev | Minimum | Maximum | P-Value |
| Pre PA* | 47.39 | 45 | 13.33 | 20 | 74 | |
| Post PA | 34.06 | 29 | 13.57 | 19 | 65 | |
| Post PA-Pre PA | -14.03 | -15 | 9.84 | -45 | 9 | <.0001 |
| Max FiO2 Rquirement Pre-Procedure % | 40.56 | 36 | 17.86 | 21 | 100 | |
| Max FiO2 Requirements 24hrs Post-Procedure-Discharge % | 28.26 | 28 | 7.63 | 21 | 52 | |
| Post-Pre | -12.30 | -7 | 15.54 | -60 | 16 | <.0001 |
| *Pulmonary Artery Systolic Pressure |